Chemistry – Atomic Structure
In this free online course, learn about atomic models, atomic orbitals and quantum numbers.
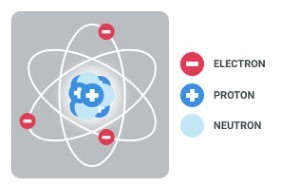
This free online course will give you an overview of atomic models and atomic theory. You will then learn some basic concepts including orbital and valency. This course will also discuss the four quantum numbers, shapes of atomic orbitals and their stability. In this course you will also look into the rules for filling electrons in orbitals which are Aufbau principle, Pauli?s exclusion principle and Hund?s rule.
What You Will Learn In This Free Course
User Reviews
Be the first to review “Chemistry – Atomic Structure”
You must be logged in to post a review.
×






There are no reviews yet.